About This Item
- Full TextFull Text(subscription required)
- Pay-Per-View PurchasePay-Per-View
Purchase Options Explain
Share This Item
The AAPG/Datapages Combined Publications Database
Journal of Sedimentary Research (SEPM)
Abstract
Differential Mobility of Elements in Burial Diagenesis of Siliciclastic Rocks
Robert P. Wintsch, Cindy M. Kvale
ABSTRACT
Mass-balance calculations involving smectite-rich siliciclastic mudstones sampled from boreholes show that significant K2O addition and CaO loss correlate strongly with the progress of illitization of smectite and with the dissolution of calcite. The calculations are based on the immobility of Al2O3, and require a chemically homogeneous sediment at deposition, such that the shallower samples represent protolith compositions of deeper samples. Constancy of ratios of TiO2/Al2O3 are used to (1) establish that both TiO2 and Al2O3 have similarly low differential mobilities, and (2) identify stratigraphic sections available in the literature that are sufficiently homogeneous to allow the calcu ations.
Our calculations show that most major-oxide components are immobile at the 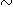 5% limit of resolution of the calculations. Exceptions are K2O, which is almost universally added to mudstones, and CaO, which is commonly removed from mudstones during diagenesis. The addition of up to 50% K2O correlates with percent illite in mixed-layer illite/smectite, indicating that illitization of smectite involves fixation of K2O from diagenetic fluids. Removal of
5% limit of resolution of the calculations. Exceptions are K2O, which is almost universally added to mudstones, and CaO, which is commonly removed from mudstones during diagenesis. The addition of up to 50% K2O correlates with percent illite in mixed-layer illite/smectite, indicating that illitization of smectite involves fixation of K2O from diagenetic fluids. Removal of 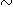 40% CaO from some sections correlates with decreasing calcite mode and is attributed to dissolution of calcite. Together these reactions lead to a 10-15% loss of mineral volume, a volume loss that must be added
to porosity loss when modeling compaction.
40% CaO from some sections correlates with decreasing calcite mode and is attributed to dissolution of calcite. Together these reactions lead to a 10-15% loss of mineral volume, a volume loss that must be added
to porosity loss when modeling compaction.
Illitization and carbonate dissolution may be linked, because K-fixation during illitization decreases the pH of the local fluid (a "reverse weathering reaction"), which enhances carbonate dissolution. Addition of up to 20% Na2O in some rocks may be caused by albitization of plagioclase, but Na2O addition is not pervasive. The apparent lack of mass transfer associated with dissolution of kaolinite and feldspars or precipitation of chlorite implies that these reactions are balanced by other reactions such that their oxide components in mudstones are conserved. Limited data from sandstone-mudstone pairs are consistent with transfer of K2O from sandstones to mudstone via rising diagenetic fluids. Except for CaO and the alkalis, diagenesis of siliciclastic mudstones appears isochemical (± 5% relative) for most major oxide components. The much larger changes recorded in some reservoir sandstones are clearly exceptions, and may not reflect changes occurring at the scale of kilometers.
Pay-Per-View Purchase Options
The article is available through a document delivery service. Explain these Purchase Options.
| Watermarked PDF Document: $14 | |
| Open PDF Document: $24 |
